Gilead's coronavirus drug remdesivir shows progress in monkeys
Animal testing for experimental COVID-19 treatment shows promising results, NIH says
Get all the latest news on coronavirus and more delivered daily to your inbox. Sign up here.
The experimental coronavirus drug remdesivir has recently shown promising results in a small study with infected monkeys, the National Institutes of Health announced Friday.
The study by government scientists in Montana found treatment with the drug, developed by Gilead Sciences, resulted in “significantly better health” for the monkeys that were treated compared to the monkeys that were untreated. Officials noted the findings have not yet faced peer review, the academic process for ensuring the validity of scientific studies.
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| GILD | GILEAD SCIENCES INC. | 134.25 | -2.63 | -1.92% |
CORONAVIRUS PATIENTS RESPONDING TO GILEAD DRUG REMDESIVIR: REPORT
Monkeys that were untreated had difficulty breathing and scientists found more damage to their lungs from COVID-19 compared to the treated animals, according to the NIH.
Officials said the data indicates treating COVID-19 patients with the drug as early as possible will help “achieve maximum treatment effect.”
Another trial conducted by the University of Chicago Medicine found most COVID-19 patients given daily treatments of remdesivir were released from the hospital in less than a week. Several other studies are ongoing.
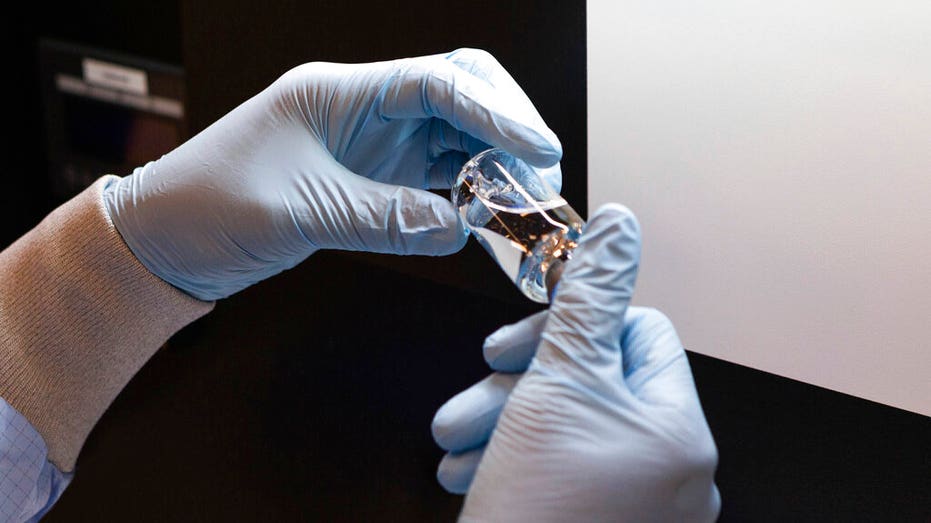
In this photo provided by Gilead Sciences, a vial of the investigational drug remdesivir is visually inspected at a Gilead manufacturing site. (Gilead Sciences via AP)
CORONAVIRUS PATIENTS RUSH TO JOIN GILEAD DRUG STUDY
Gilead said this week it looks “forward to data from ongoing studies becoming available.”
There were more than 2.2 million COVID-19 cases worldwide as of Friday, including more than 683,000 in the U.S. The virus has killed more than 150,000 people globally.
President Trump has also pointed to the drug as a possible treatment for the coronavirus, saying last month that it “seems to have a very good potential.”

Given through an IV, the medication is designed to interfere with an enzyme that reproduces viral genetic material. (Gilead Sciences via AP)
GET FOX BUSINESS ON THE GO BY CLICKING HERE
Gilead’s stock has seen a boost from all the positive talk around the drug. The company announced Friday it would release first-quarter financial results on April 30.
The company previously said it had enough doses for up to 140,000 treatment courses and that it was aiming to make 500,000 by October and more than 1 million by the end of 2020, the Associated Press reported.
FOX Business' Lucas Manfredi contributed to this report.
CLICK HERE TO READ MORE ON FOX BUSINESS