Coronavirus patients responding to Gilead drug Remdesivir: Report
Shares of Gilead Sciences were up more than 14 percent in after-hours trading
Trump lists companies helping produce PPE, treatments
President Trump gives an update on the latest potential coronavirus treatments and the companies producing personal protection equipment to combat coronavirus.
Shares of Gilead Sciences were up more than 8 percent in premarket trading on Friday after a report by STAT that a recent clinical trial of Gilead Sciences' Remdesivir, an antiviral medicine used as a potential treatment for Ebola, found rapid recoveries in coronavirus patients' fever and respiratory symptoms.
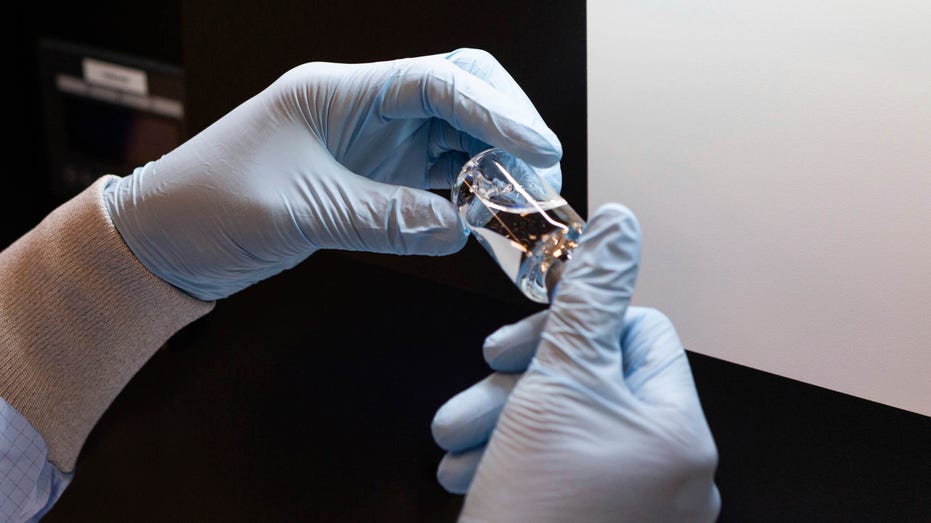
In this March 2020 photo provided by Gilead Sciences, a vial of the investigational drug remdesivir is visually inspected at a Gilead manufacturing site in the United States. Given through an IV, the medication is designed to interfere with an enzyme
The trial, conducted by the University of Chicago Medicine, found nearly all patients who were given daily infusions of Remdesivir were discharged from the hospital in less than a week. The university recruited 125 coronavirus patients, 113 of which had "severe disease" into two clinical trials to assess five and 10-day treatment courses of the drug.
BLOOD PURIFICATION DEVICE FOR CORONAVIRUS PATIENTS GRANTED EMERGENCY FDA APPROVAL: CYTOSORBENTS CEO
Kathleen Mullane, the infectious disease specialist overseeing the University of Chicago's Remdesivir studies, told STAT that the results have been promising so far.
"Most of our patients are severe and most of them are leaving at six days, so that tells us the duration of therapy doesn’t have to be 10 days," Mullane said. "We have very few that went out to 10 days, maybe three."
Although a fever is no longer a requirement for people to go into clinical trials at the hopsital, Mullane said that when patients do come in with high fevers, "they do [reduce] quite quickly."
"We have seen people come off ventilators a day after starting therapy," she added. "So, in that realm, overall our patients have done very well."
While encouraged by the University of Chicago data, Mullane warned not to draw any conclusions about the drug's effectiveness.
"It’s always hard," she said, because the severe trial doesn’t include a placebo group for comparison. "But certainly when we start [the] drug, we see fever curves falling."
CORONAVIRUS SURVIVORS TELL TRUMP HYDROXYCHLOROQUINE HELPED
Last month, President Trump touted the drug's potential, saying it "seems to have a very good result."
Remedsivir was one of the first medicines identified as having the potential to treat coronavirus in lab tests. If deemed safe and effective in clinical trials, it could lead to fast approval from the Food and Drug Administration and become the first approved treatment against the disease.
While the results of Gilead's trial offer a glimpse of the drug's effectiveness, it has not been deemed to be a potential coronavirus treatment due to an overall lack of data.
According to STAT, two Remdesivir studies in China had enrollment suspended partway through because there were not enough patients available, and a recent report of patients given the drug under a special program to make it available to those who are very ill generated "both excitement and skepticism."
GET FOX BUSINESS ON THE GO BY CLICKING HERE
In a statement Thursday, Gilead said, "What we can say at this stage is that we look forward to data from ongoing studies becoming available."
The company is expecting results from two other ongoing studies soon, one of which includes 2,400 coronavirus patients with "severe disease" from 152 different clinical trials sites around the world, and another including 1,600 patients with "moderate disease" at 169 clinical trial sites around the world.
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| GILD | GILEAD SCIENCES INC. | 132.06 | -0.99 | -0.74% |
Gilead Sciences' stock closed at $76.54 per share at the end of Thursday's trading session.
CLICK HERE TO READ MORE ON FOX BUSINESS





















