Lilly starts late-stage study of coronavirus drug in nursing homes
Seniors are among the most affected by coronavirus pandemic
(Reuters) - U.S. drugmaker Eli Lilly & Co said on Monday it is beginning a late-stage trial to study whether one of its experimental COVID-19 antibody treatments can prevent the virus’ spread in residents and staff in U.S. nursing homes.
The phase 3 trial will test LY-CoV555, a treatment developed in partnership with Canadian biotech AbCellera, is expected to enroll up to 2,400 participants who live or work at a facility that have had a recently diagnosed case of COVID-19.
J&J BEGINS CLINICAL TRIAL OF SINGLE-DOSE CORONAVIRUS VACCINE
“COVID-19 has had a devastating impact on nursing home residents,” Lilly’s chief scientific officer Daniel Skovronsky said in a statement. “We’re working as fast as we can to create medicines that might stop the spread of the virus to these vulnerable individuals.”
Lilly is already testing the drug in hospitals to study whether it can work as a treatment in patients who have the disease. This trial will test whether it works prophylactically.
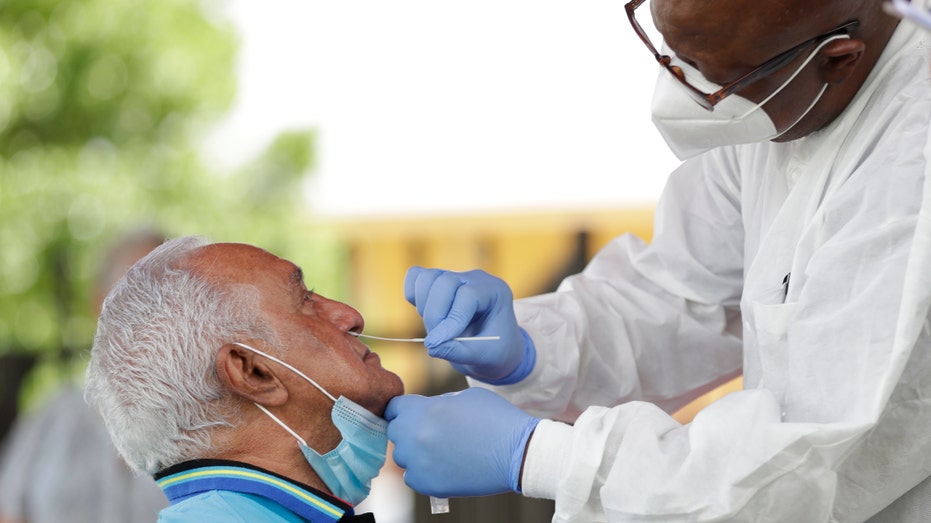
Lab technician James Donald, right, uses a nasal swab to test Hugo Marti for COVID-19, Tuesday, July 28, 2020, at the AHEPA Apartments in Miami. (AP Photo/Wilfredo Lee)
It is launching the phase 3 trial in partnership with several long-term care facility networks across the country as well as the U.S. National Institute of Allergy and Infectious Diseases (NIAID).
Lilly said in order to speed the study it has created mobile research units including retrofitted recreational vehicles that can be deployed in response to outbreaks of the virus at nursing homes across the U.S.
CLICK HERE TO READ MORE ON FOX BUSINESS
LY-CoV555 belongs to a class of treatments known as monoclonal antibodies that are among the most widely used biotechnology medicines. Regeneron Pharmaceuticals Inc and other drugmakers are testing similar treatments against COVID-19.
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| LLY | ELI LILLY & CO. | 989.81 | +22.84 | +2.36% |
Last week Lilly told investors that LY-CoV555 had moved into mid-stage trials as a treatment and would start late stage-trials in the coming weeks. It expects efficacy data from the mid-stage trial in the fourth quarter.




















