Saniderm Advanced Hand Sanitizer recalled over potential toxic chemicals
FDA had warned about 9 sanitizers made by Mexico-based Eskbiochem SA de CV due to possible methanol
UVT Inc. is voluntarily recalling more than 38,000 liters of Saniderm Advanced Hand Sanitizer due to the potential presence of methanol, according to an announcement posted by the Food and Drug Administration on Monday.
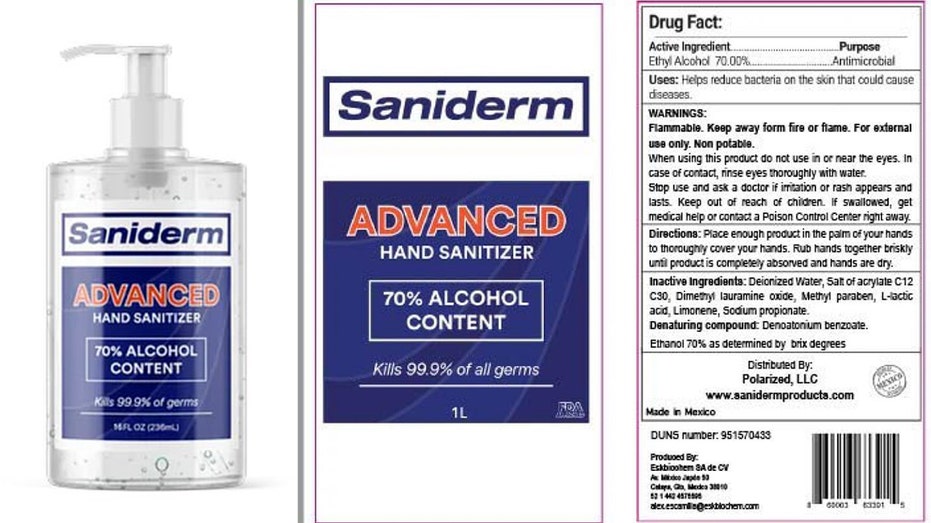
Saniderm Advanced Hand Sanitizer (Food and Drug Administration)
The affected hand sanitizer includes lot number 0530 and an expiration date of 04/2022. The company said it is notifying its distributors and customers by phone calls, emails and letters to arrange for replacement and destruction of the recalled products, according to the announcement.
The company recommends that all retailers and distributors of the product issue their own recall, and consumers with the recalled hand sanitizer should stop using it and return the product to its place of purchase.
To date, UVT Inc. has not received any reports of adverse effects associated with the recall. UVT Inc. did not immediately return FOX Business' request for comment.
FDA TELLS HAND SANITIZER PRODUCERS TO MAKE IT UNPALATABLE AFTER SURGE IN POISON CONTROL CALLS
According to the FDA, methanol can be toxic when absorbed through the skin or ingested.
"Although all persons using these products on their hands are at risk, young children who accidently [sic] ingest these products and adolescents and adults who drink these products as an alcohol (ethanol) substitute, are most at risk for methanol poisoning," the company said.
Substantial methanol exposure can result in nausea, vomiting, headache, blurred vision, permanent blindness, seizures, coma, permanent damage to the nervous system or death.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
Saniderm Advanced Hand Sanitizer is among nine products manufactured by Mexico-based company Eskbiochem SA de CV that received a warning from the FDA last week for the potential risk of methanol poisoning.
Other Eskbiochem products that received the FDA's warning are All-Clean Hand Sanitizer, Esk Biochem Hand Sanitizer, CleanCare NoGerm Advanced Hand Sanitizer 75% Alcohol, Lavar 70 Gel Hand Sanitizer , The Good Gel Antibacterial Gel Hand Sanitizer, CleanCare NoGerm Advanced Hand Sanitizer 80% Alcohol, CleanCare NoGerm Advanced Hand Sanitizer 75% Alcohol and CleanCare NoGerm Advanced Hand Sanitizer 80% Alcohol.
On June 17, the FDA contacted Eskbiochem recommending the company remove the "potentially dangerous" hand sanitizer products from the market. As of June 19, the agency said Eskbiochem has not taken any action to do so and was not aware of any reports of adverse effects associated with any of the recalled hand sanitizer products.
Eskbiochem did not immediately return FOX Business' request for comment on the FDA's letter.
The FDA says that anyone who has been exposed to hand sanitizer products with methanol should seek immediate treatment and dispose of the product immediately in "appropriate hazardous waste containers."




















