Trump launches 'Operation Warp Speed' for coronavirus vaccine
Operation Warp Speed is 'a massive scientific industrial and logistical endeavor'
President Trump on Friday announced "Operation Warp Speed" -- a new initiative that is aimed at developing, manufacturing and distributing a "proven" coronavirus vaccine.
Trump described the administration's plan as "a massive scientific industrial and logistical endeavor unlike anything our country has seen since the Manhattan Project" of World War II, with the intent to rapidly develop and distribute a vaccine with help from the U.S. military and world-renowned doctors and scientists.
"We'd love to see if we can do it prior to the end of the year," the president said. "We think we're going to have some very good results coming out very quickly."

President Donald Trump speaks about the coronavirus in the Rose Garden of the White House, Friday, May 15, 2020, in Washington. (AP Photo/Alex Brandon)
Get all the latest news on coronavirus and more delivered daily to your inbox. Sign up here.
The administration is providing roughly $10 billion through the Coronavirus Aid, Relief, and Economic Security (CARES) Act to support the medical research effort, including funding for vaccine development, therapeutics and diagnostics.
LARGE SCALE TESTING EXPECTED BY JULY: NIH SECRETARY
Dr. Moncef Slaoui, an immunologist who has helped create five major novel vaccines within the private sector, and Gen. Gustave Perna, a four-star general who oversees 190,000 service members, civilians and contractors in the U.S. Army Materiel Command, will lead Operation Warp Speed.
Trump added that the administration has already looked at 100 vaccine candidates "from all over the world," and has narrowed those options down to 14 in recent weeks.
TRUMP: CORONAVIRUS VACCINE COULD COME BY THE END OF THE YEAR
"There's never been a vaccine project like this anywhere in the world," he said.
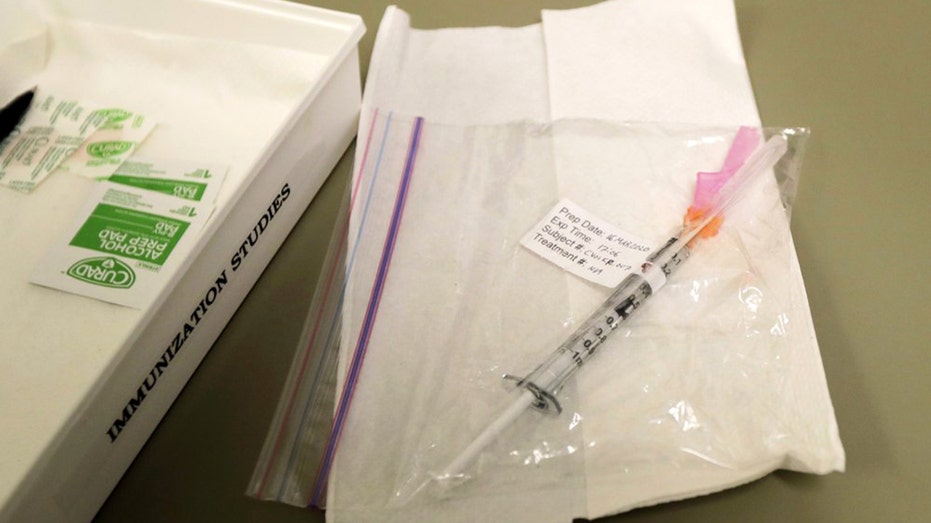
A syringe containing the first shot given in the first-stage safety study clinical trial of a potential vaccine for COVID-19, the disease caused by the new coronavirus, rests on a table, Monday, March 16, 2020, at the Kaiser Permanente Washington Hea
The operation will include efforts from Health and Human Services; Centers for Disease Control and Prevention; Food and Drug Administration; National Institutes of Health; the Biomedical Advanced Research and Development Authority; the Department of Defense; private firms; and other federal agencies, including the Department of Agriculture, the Department of Energy and the Department of Veterans Affairs, according to a press release.
HHS Secretary Alex Azar called the operation "one of the greatest scientific and humanitarian accomplishments in history."
GET FOX BUSINESS ON THE GO BY CLICKING HERE
"Dr. Slaoui and General Perna are ideal leaders for this unprecedented effort to get vaccines, therapeutics, and diagnostics to American patients much faster than ever before," Azar said. "Since January, America’s scientists and innovators have been working day and night on this national effort. President Trump has refused to accept business-as-usual timelines for vaccines and other essential tools, and instead has insisted that America, and the world, needs answers faster. Under the president’s leadership, his administration and American industry will squeeze every last inefficiency out of the process and pour every resource we can into this effort."




















