Could this Alzheimer’s prevention drug prevent onset of the disease?
Biogen, which researches treatments for neurological disorders, is pursuing regulatory approval for its Alzheimer’s treatment, aducanumab, from the U.S. Food and Drug Administration.
Fox News medical correspondent Dr. Marc Siegel said on “Varney & Co.” he believes this drug could help prevent the onset of Alzheimer’s disease.
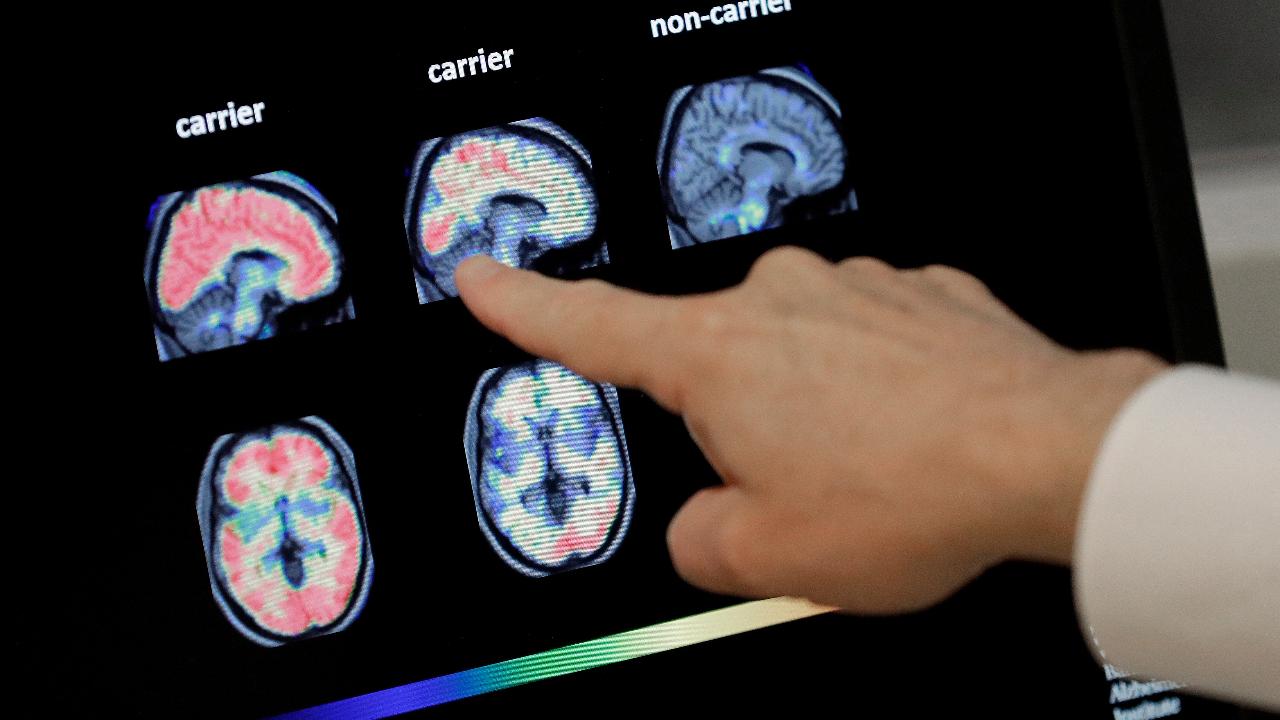
Alzheimer’s is caused by the buildup of an abnormal protein in the brain which interferes with brain cell communication, Siegel said.
“So [the] question is if the proteins aren’t there clunking up your brain, can we prevent them from forming?” he asked. “[Anducanumab is] a targeted therapy. It’s an antibody you inject ... that goes right for that protein,” Siegel said.
A Biogen study of the drug resulted in improvements for patients on measures of cognition and function such as memory, orientation, and language, according to Biogen. Patients also showed improvement on daily tasks including conducting personal finances, performing household chores such as cleaning, shopping, and doing laundry, and independently traveling out of the home, according to the company.
Biogen's study is the first time scientists have been able to show that clearing the aggregated amyloid beta, the abnormal protein, showed improvements in test patients, Dr. Anton Porsteinsson, the lead Biogen investigator, said in a prepared statement.
Biogen's CEO Michel Vounatsos said they're hopeful this drug will reduce Alzheimer's effects on the brain.
“This is the result of groundbreaking research and is a testament to Biogen’s steadfast determination to follow the science and do the right thing for patients,” Michel Vounatsos, Biogen CEO said in a prepared statement. “We are hopeful about the prospect of offering patients the first therapy to reduce the clinical decline of Alzheimer’s disease and the potential implication of these results for similar approaches targeting amyloid beta.”
According to the Centers for Disease Control, as many as 5 million Americans were living with Alzheimer's disease in 2014, a number projected to nearly triple to 14 million by the year 2060.
GET FOX BUSINESS ON THE GO BY CLICKING HERE