White House announces $750M deal for Abbott Labs' COVID-19 rapid test
Through Operation Warp Speed and in partnership with Abbott Laboratories, production of the test will be ramped up to 50 million tests monthly.
Abbott’s rapid coronavirus test is ‘huge game changer’: Dr. Marc Siegel
Fox News medical correspondent Dr. Marc Siegel on Abbott’s new rapid coronavirus test.
The White House has announced a $750 million deal with Abbott Laboratories Thursday to buy 150 million of its new rapid coronavirus tests, the BinaxNOW COVID-19 Ag Card.
"This is a major development that will help save more lives by further protecting America’s most vulnerable and allow our country to remain open, get Americans back to work, and get kids back to school," the White House said in a statement. "Under President Trump’s leadership, the United States is leading the world in testing and the development of a wide range of reliable tests."
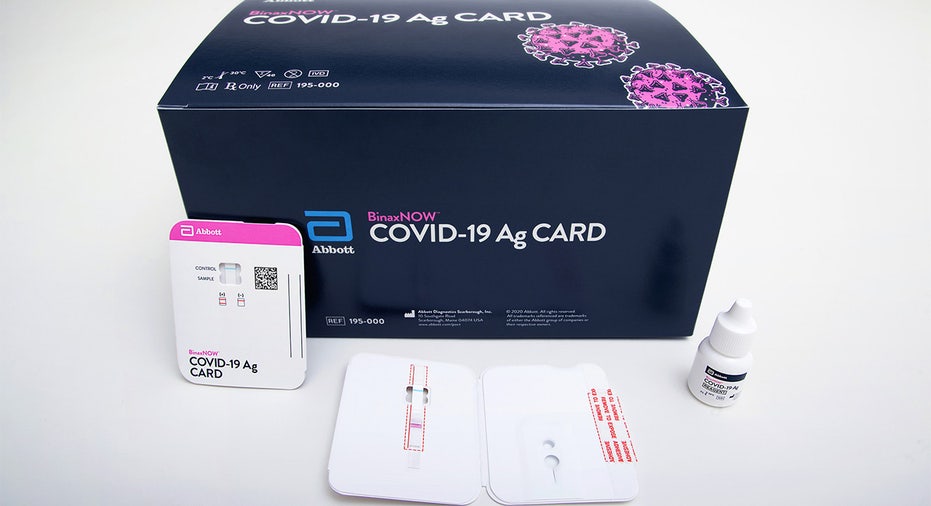
The $5 test, which is roughly the size of a credit card, is administered by a healthcare professional using a nasal swab.
FDA GREENLIGHTS ABBOTT'S NEW $5 CORONAVIRUS TEST THAT PROVIDES RESULTS WITHIN 15 MINUTES
BinaxNow uses lateral flow technology, similar to an at-home pregnancy test, to search for virus proteins and is intended to be used for patients within seven days of feeling coronavirus symptoms. The test can provide results in up to 15 minutes.
Abbott's BinaxNOW™ COVID-19 Ag Card is a rapid, reliable and affordable tool for detecting active coronavirus infections at massive scale. (Photo courtesy of Abbott Laboratories)
According to Abbott, the test has a demonstrated sensitivty, or the percent of positive cases that it can accurately detect, of about 97%.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
Using a free app called NAVICA, those who are looking to return to work or school will be able to display a digital health pass via a QR code that shows their negative test result.

NAVICA™ is a no-charge complementary phone app, which allows people to display their BinaxNOW test results. (Photo courtesy of Abbott Laboratories)
Through Operation Warp Speed and in partnership with Abbott Laboratories, production of the test will be ramped up to 50 million tests monthly.
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| ABT | ABBOTT LABORATORIES | 82.56 | -1.74 | -2.06% |
BinaxNOW is the sixth test that Abbott has launched in the U.S. to help fight the coronavirus pandemic. To date, the company has provided more than 27 million COVID-19 tests in the United States, including 14 million detection tests and 13 million antibody tests.
Abbott’ stock closed at $111.29 per share, surging nearly 8 percent at the end of Thursday’s trading session.