Complaint letter filed with SEC regarding Natera’s promotion of prenatal screening technology
Natera and other companies were the subject of a recent report on potential pitfalls in prenatal screening
FIRST ON FOX: A tech watchdog has filed a complaint letter with the Securities and Exchange Commission (SEC), alleging that biotech firm Natera misled the public about its prenatal screening technology.
Submitted on Friday, the complaint comes roughly two weeks after the New York Times published a story on the accuracy of non-invasive prenatal tests (NIPTs) from companies like Natera. That report, the Campaign for Accountability (CfA) contends, pointed to data that purportedly told a different story than what Natera's marketing, SEC filings and public statements did.
Natera told FOX Business that CfA's allegations were bogus: "The entire premise of this story and every allegation in it originates from meritless, opportunistic legal maneuvering from plaintiffs firms trying to foment litigation. … If any of these purported claims are ever brought, we look forward to vigorously defending ourselves and exposing these claims for what they are through the proper legal channels."
CfA's complaint points to earnings calls in which founder Matthew Rabinowitz compared the artificial intelligence for its test, known as Panorama, to the supercomputer that beat chess champion Garry Kasparov. That and other purportedly "extravagant claims" from Natera "are, quite simply, untrue," according to CfA. Another aspect of the complaint is Chief Operating Officer (COO) Robert Allan Schueren selling about 10,800 shares of stock in the weeks leading up to the Times story.
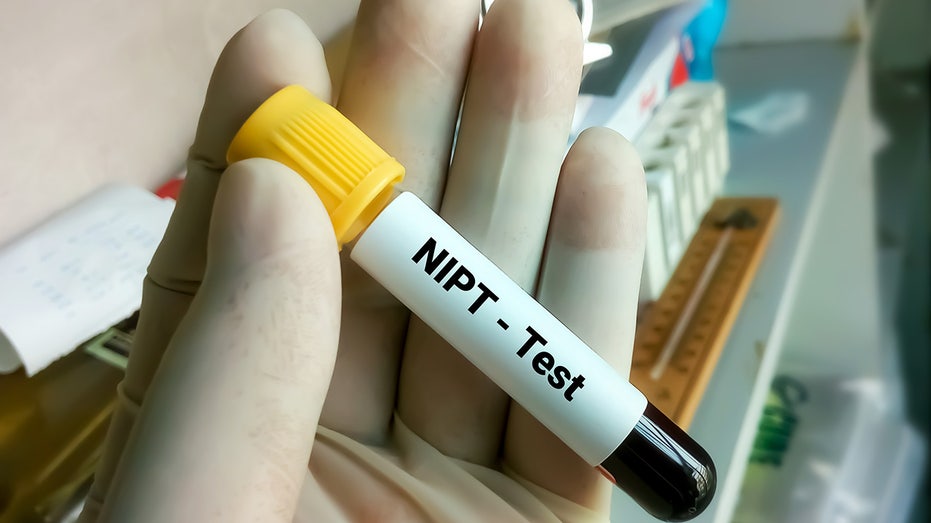
Blood sample for NIPT or non-invasive prenatal testing, diagnosis for fetal Down syndrome during pregnancy. (iStock / iStock)
CfA also referenced to a Times graphic that it said was later edited. That graphic claimed that a Natera study suggested a high number of false positives for its NIPT for DiGeorge syndrome, which is associated with intellectual disability and heart defects.
"In a recent study, Natera said that its latest algorithm would identify about an equal number of false positives," the Times wrote. "But that same study also included the results from when the tests were actually taken. Those numbers suggest there would be three times as many false positives as actual cases."
CfA's filing hones in on Panorama's patient brochure, website and SEC filings — arguing that they either omit or don't provide enough information on the company's false positives.
A woman receiving an ultrasound. (iStock)
"By disingenuously touting the accuracy of its NIPT to investors, Natera may be directly or indirectly, in the offer or sale of securities, by use of the means or instruments of transportation or communication in interstate commerce or by the use of mails, obtaining money or property by means of untrue statements of material fact, or omitting to state a material fact necessary in order to make the statements made in light of the circumstances under which they were made, not misleading," CfA's filing alleges.
Natera filings have provided general statements about the uncertainty, and its most recent 8-K discusses the prevalence of certain microdeletions in the population.
It also says that its testing is accurate in more than 99% of cases. Its SMART study, which Rabinowitz cited during his earnings call, "reported clinical results for 22q11.2 (22q) deletion syndrome to 18,014 patients, and we delivered a correct result to 17,982 of those patients (99.8%)."
FLORIDA ABORTION LEGISLATION WOULD BAN PROCEDURE AFTER 15 WEEKS
"Our results are even stronger with our new Panorama AI algorithm. In addition, we were able to successfully detect 75% of 22q deletions in the study, and 83% of cases with our improved algorithm."
The controversy over NIPTs has prompted intense debate as the method can alter expectations for pregnant mothers and potentially influence a decision to abort the fetus. Fox News Digital reported last week that multiple GOP politicians called for some form of accountability after the Times' analysis, which found an 85% false positive rate for the five most common microdeletion tests from multiple companies. The Times article was also cited by multiple law firms in announcing investigations of Natera.

Pregnant mother touching and looking at her pregnant belly at home. (iStock)
In studying NIPT, researchers also look at the "positive predictive value" (PPV), which refers to the likelihood that the result is a true positive and the disease is actually present in the population.
Dr. Tara Sander Lee, a researcher with the anti-abortion Charlotte Lozier Insitute, told FOX Business while NIPTs are a lot better than standard screens, they continue to face issues with false positives among low-risk women. For example, a 2014 study found a PPV of 45.5% in screening for Down Syndrome in low risk women. That PPV, she said, means "the probability that a woman is a true positive is less than 50%. Over half of results will be wrong and [the] disease will not be present."
In a press release earlier this month, Natera said its microdeletion PPVs have ranged from about 5% to 50% and compared its PPVs to historic standards for prenatal screening.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
"While a false positive result can be difficult information for expectant families to receive, these PPVs compare favorably to historically accepted maternal serum based prenatal screening methods, which were used routinely on ~80% of all pregnancies for the past 40 years and are still recommended by ACOG [the American College of Obstetricians Gynecologists] today," the company said.
"In fact, historically accepted prenatal screening tests have a PPV around 3-5% (1/33-1/20) for chromosomal conditions. In contrast, Natera’s microdeletion test has PPVs ranging from ~5% to 50%, depending on the specific microdeletion syndrome being screened."
Screenings are not necessarily the end of the road for pregnant women looking to uncover more about their unborn children.
In line with ACOG, Natera says it recommends diagnostic testing and genetic counseling.
CLICK HERE TO READ MORE ON FOX BUSINESS
"We require a licensed physician to order tests, and we don’t perform testing unless a physician signs the requisition indicating that the patient has consented to testing," the company said in its press release.
"In addition, we put the PPV for each condition directly onto the patient report with a recommendation for confirmatory diagnostic testing, so physicians can discuss the results with their patients."




















