Which companies are leading the coronavirus vaccine race?
Bill Gates is most optimistic about these three companies
Companies based in the U.S. and abroad are competing for funds to develop a coronavirus vaccine as soon as possible.
MELINDA GATES: THESE PEOPLE DESERVE TO GET CORONAVIRUS VACCINE FIRST
Experts say developing such a vaccine can take years and billions of dollars, but already vaccine testing is underway in countries including the United States and South Korea.
Billionaire Bill Gates, who has contributed millions to the fights against the virus, said in June that he's most encouraged by the three vaccine candidates made by Moderna, AstraZeneca and Johnson & Johnson. Gates believes it is possible to scale up to 10 billion doses of a successful vaccine in two years in order to achieve global herd immunity, he said according to Forbes.
A researcher at Protein Sciences moves a vial in a lab, Thursday, March 12, 2020, in Meriden, Conn. (AP Photo/Jessica Hill)
There are 172 COVID-19 vaccines in development and 13 in clinical testing, according to tracking by the Milken Institute.
Here are some of the companies furthest ahead in pursuing coronavirus prevention:
Moderna
Massachusetts-based Moderna Therapeutics was the first U.S. company to get a coronavirus vaccine ready for human testing. Its CEO said the company would put the vaccine at the same price point as similar existing vaccines.
Moderna is set to test its vaccine on 30,000 volunteers starting in July.
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| MRNA | MODERNA INC. | 54.35 | +5.81 | +11.97% |
| AZN | ASTRAZENECA PLC | 182.85 | +0.33 | +0.18% |
| JNJ | JOHNSON & JOHNSON | 221.32 | -1.19 | -0.53% |
AstraZeneca
United Kingdom-based AstraZeneca is partnering with Oxford University to deliver a coronavirus vaccine to European countries including France and Germany should the vaccine be proven effective.
Testing began in April.
WHY IS BILL GATES IN THE FIGHT AGAINST CORONAVIRUS?
Johnson & Johnson
Johnson & Johnson's pharmaceutical company Janssen worked with Beth Israel Deaconess Medical Center, part of Harvard Medical School, to test and select its lead coronavirus vaccine candidate in March.
Johnson & Johnson plans to start human clinical studies in July.
The company has partnered with the U.S. Biomedical Advanced Research and Development Authority, which is part of the Department of Health and Human Services, to commit to investing more than $1 billion in fighting the coronavirus.
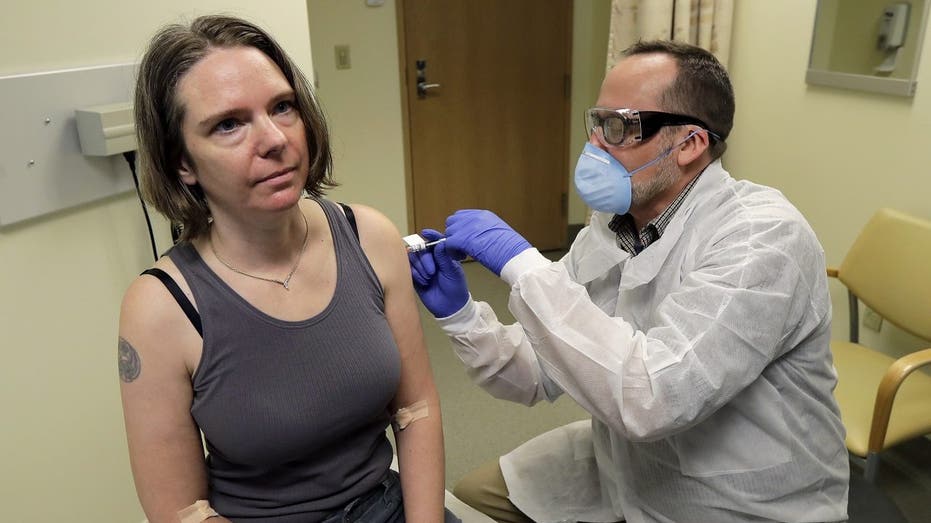
Jennifer Haller, left, receives the first shot in the first-stage safety study for a potential COVID-19 vaccine for at the Kaiser Permanente Washington Health Research Institute in Seattle. (AP Photo/Ted S. Warren)
Inovio
Inovio Pharmaceuticals is a Pennsylvania-based biotech company that received a $71 million grant from the Department of Defense on June 23 to scale up its Cellectra intradermal DNA vaccine delivery device.
Inovio already received $10 million from billionaire Bill Gates' Coalition for Epidemic Preparedness Innovations to accelerate testing of the device.
Inovio said it created a coronavirus vaccine hours after getting access to the genetic sequence in mid-January.
CLICK HERE TO READ MORE ON FOX BUSINESS
CanSino
China-based CanSino received the go-ahead to begin human clinical testing in China. CanSino is known for creating the first Ebola vaccine to gain approval anywhere in the world.
The company released data from its first round of testing, which it said warranted "further investigation," in late May, BioSpace reported.




















