Roche ramping up coronavirus antibody test production to 'upper-digit millions': CEO
Swiss company's antibody test appears more effective than its competitors'
Get all the latest news on coronavirus and more delivered daily to your inbox. Sign up here.
Roche CEO Severin Schwan said his company will ramp up production of its new coronavirus antibody test to "the high upper-digit millions" in May.
CORONAVIRUS PATIENT STUDY RAISES QUESTIONS ABOUT WHETHER TEMPERATURE CHECKS WORK
"In May alone we will be able to ramp up production of this antibody test to the high upper-digit millions, and we will further increase capacity for the coming months," Schwan said.
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| RHHBY | ROCHE HOLDING AG | 51.09 | +0.51 | +1.00% |
The Swiss company's antibody test is not the first to received FDA Emergency Use Authorization, but it appears to be the most effective compared to its competitors from Cellex and Premier Biotech. Roche's test has a sensitivity of 100 percent and a specificity of 99.8 percent.

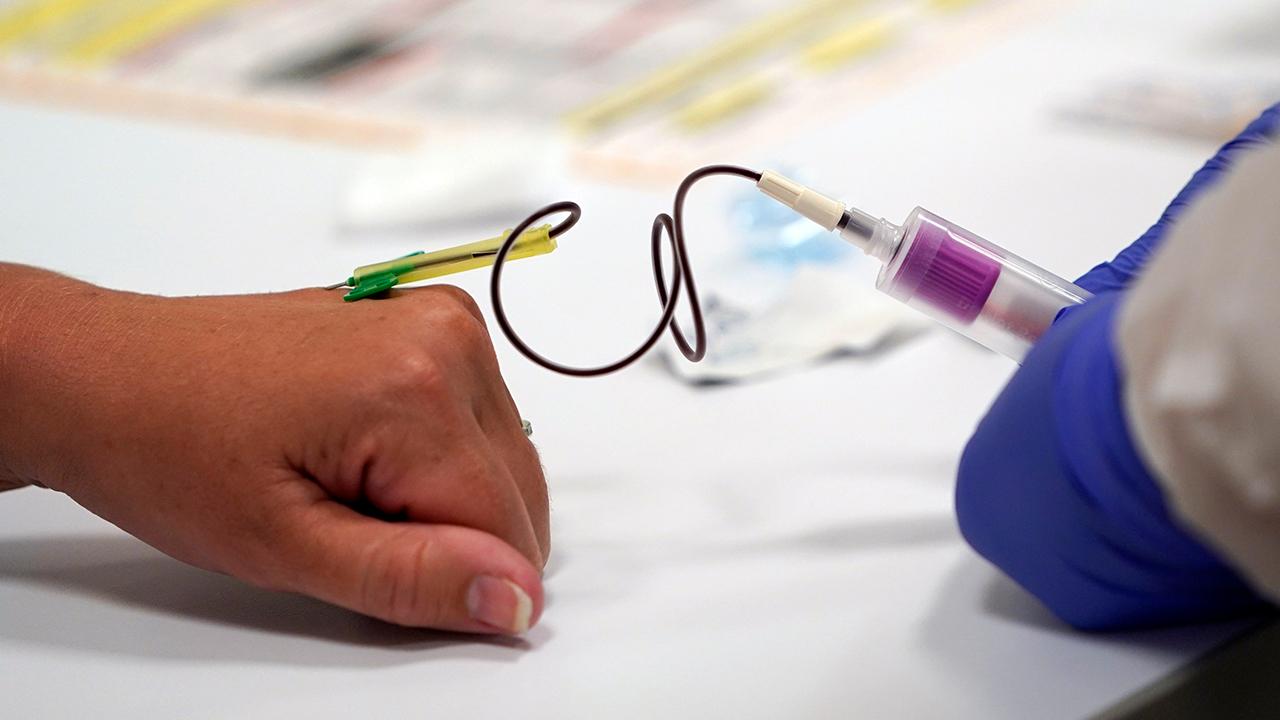
A woman's blood is collected for testing of coronavirus antibodies at a drive-thru testing site in Hempstead, N.Y., April 14, 2020. (AP Photo/Seth Wenig)
Antibody testing can also help researchers learn whether patients can be reinfected with the novel coronavirus.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
"There's a high likelihood after the infection of this coronavirus that you will acquire immunity, but this still needs to be proven by the studies where you follow patients who have been infected," Schwan said. "One of the benefits of this antibody test is that you can follow up such patients over time and see whether they get reinfected or not, but there's a good chance, like with other coronaviruses, that you get immune after you have been infected."