Coronavirus convalescent plasma treatment 'appears to be safe': FDA commissioner
According to Hahn, more than 28,000 Americans have received convalescent plasma treatment
During an appearance on ABC "This Week," Food and Drug Administration Commissioner Stephen Hahn said the agency is looking into the effectiveness of convalescent plasma as a potential treatment for the coronavirus.
Convalescent plasma is the liquid portion of a person's blood that is collected from patients who have recovered from COVID-19, which contains antibodies that help support immunity and protect from future infections, according to the FDA.
"We're looking at whether that's effective or not," Hahn said. "It appears to be safe."
Convalescent plasma therapy has a very low risk of allergic reactions, lung damage and difficulty breathing, and transmission of infections, including IV and hepatitis B and C, according to the Mayo Clinic.
WHICH COUNTRY WILL DEVELOP CORONAVIRUS VACCINE FIRST?
Hahn noted more than 28,000 Americans have received convalescent plasma treatment, and encouraged anyone who has recovered from the coronavirus to contact their local Red Cross about donating plasma.
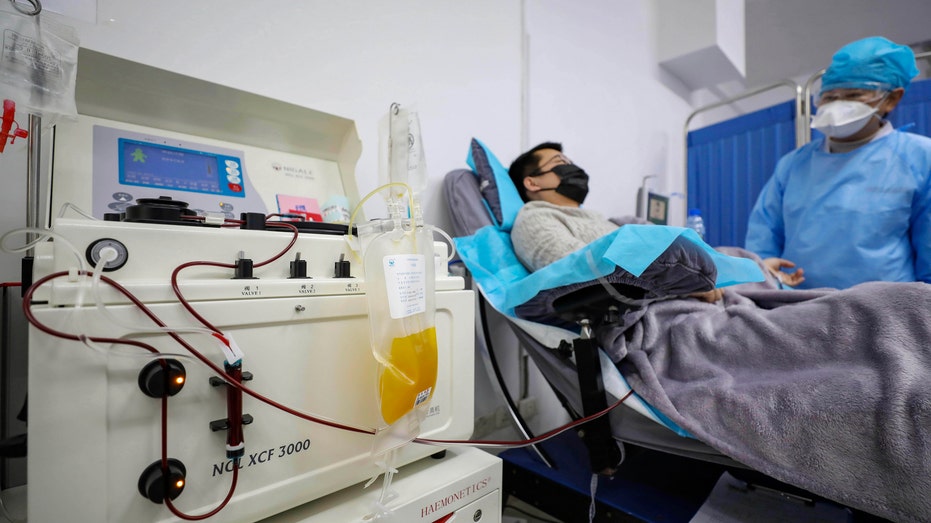
Dr. Kong Yuefeng, a recovered COVID-19 patient who has passed his 14-day quarantine, donates plasma in the city's blood center in Wuhan in central China's Hubei province Feb. 18, 2020. (Chinatopix via AP)
"If you've had COVID-19, you have an opportunity to give back by contacting your Red Cross, by contacting a local plasma center and donating," Hahn said. "It could save a life."
Those interested in donating can fill out a form on the Red Cross website. According to the Red Cross, eligible donors will receive an appointment to donate at a Red Cross or non-Red Cross collection site and may give convalescent plasma every 28 days.
BELLAGIO ERROR MAY BE BIGGEST SPORTSBOOK LOSS FOR VEGAS
Eligible donors must be at least 17 years old and weigh 110 pounds, must be in good health, and must be symptom-free and full-recovered from the coronavirus after having a prior, verified diagnosis.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
According to the latest update Johns Hopkins University, there are more than 2.8 million confirmed coronavirus cases and more than 192,000 deaths in the United States. More than 894,000 coronavirus patients have fully recovered.
CLICK HERE TO READ MORE ON FOX BUSINESS