Biden admin provides $11M for monkeypox, smallpox vaccine production
Jynneos vaccine production at a Michigan facility is expected later in 2022
The Department of Health and Human Services (HHS) is providing approximately $11 million to support the first U.S.-based fill and finish manufacturing of the vaccine used to prevent smallpox and monkeypox.
The agreement between Grand River Aseptic Manufacturing (GRAM) in Grand Rapids, Michigan; the HHS Administration for Strategic Preparedness and Response; and the Biomedical Advanced Research and Development Authority aids the company in accelerating the fill and finish manufacturing qualification and production of the Jynneos vaccine in its recently expanded facility.
The money will allow GRAM — where the vaccine will be manufactured — to purchase additional equipment necessary for the vaccine's production and to recruit and train additional staff.
Jynneos production is expected later this year.
MET OPERA DROPS VACCINE REQUIREMENT, STILL MANDATES MASKS
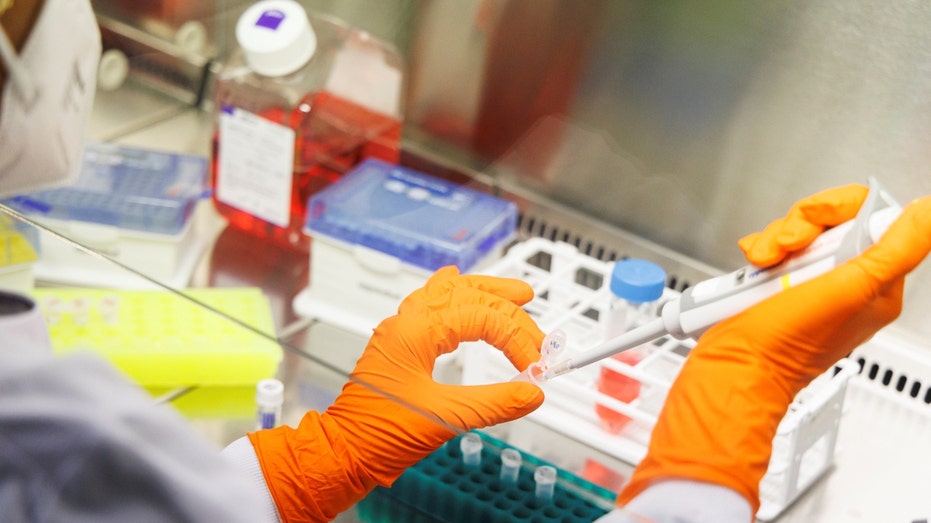
An employee of the vaccine company Bavarian Nordic works in a company laboratory in Martinsried near Munich, Germany, May 24, 2022. (Reuters/Lukas Barth / Reuters)
"We continue to build on our efforts to secure and make safe and effective vaccines readily available," HHS Secretary Xavier Becerra said in a statement. "This new agreement solidifies a domestic manufacturing capability that will bring us more vaccine sooner to end this outbreak."
Earlier this month, the maker of Jynneos, Bavarian Nordic, and GRAM announced an agreement to establish the first U.S.-based fill and finish capability for the Jynneos vaccine.

An employee of the vaccine company Bavarian Nordic works in a company laboratory May 24, 2022. (Reuters/Lukas Barth / Reuters)
GET FOX BUSINESS ON THE GO BY CLICKING HERE
The agreement was facilitated by BARDA.

Bavarian Nordic's laboratory in Martinsried near Munich, Germany, May 24, 2022. (REUTERS/Lukas Barth / Reuters)
In July, BARDA placed orders for the fill, finish and delivery of 5 million Jynneos vials from U.S. government-owned bulk vaccine stored in Denmark.
CLICK HERE TO READ MORE ON FOX BUSINESS
In that contract, Bavarian Nordic agreed to a complete a technology transfer allowing a U.S.-based contract manufacturer to fill and finish 2.5 million vials.





















