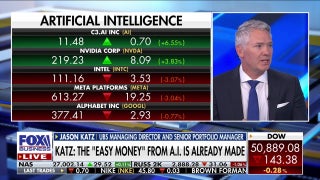
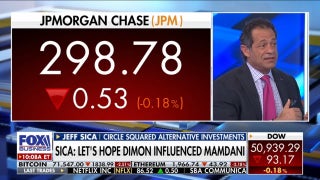
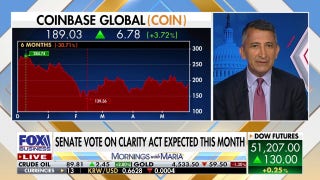
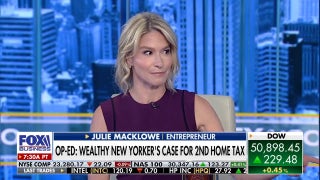
Amgen: Study shows its biosimilar version of popular arthritis drug Humira about as effective
THOUSAND OAKS, Calif. – Biologic drugmaker Amgen says its biosimilar version of rheumatoid arthritis drug Humira worked similarly in a key head-to-head test.
Amgen Inc. says its ABP 501 produced similar results in the percentage of patients in the 26-week, late-stage study who saw improvements in pain, ability to function and number of swollen and tender joints. Half the 526 participants got AbbVie Inc.'s Humira, which had sales of $12.5 billion last year. Half received ABP 501.
Amgen, based in Thousand Oaks, California, is developing nine biosimilars — drugs that are similar, but not identical to, brand-name biologic drugs. Those are grown in living cells, rather than made by mixing chemicals.
Amgen expects to launch five biosimilars between 2017 and 2019. ABP 501 could be the first for which Amgen seeks marketing approval.