Pfizer, BioNTech begin coronavirus vaccine trials in kids, publish early-stage data
The company plans to vaccinate kids as young as 12, officials said
While this week brought along pauses in coronavirus trials for Johnson and Johnson’s vaccine candidate and Eli Lilly’s antibody treatment over safety concerns,
Stocks In This Article:
Pfizer and BioNTech published early-stage data of a vaccine candidate now in Phase 3, and marked the first company in the U.S. to begin COVID-19 vaccination trials in teens.
Pfizer chose its vaccine candidate currently being tested in global late-stage trials from four candidates earlier in the pandemic. Phase 1 trial data between two candidates helped drive this decision, which revealed “a favorable balance of reactogenicity and immunogenicity” in the BNT162b2 candidate over BNT162b1, per the study.
Findings from the early-stage trial were published on Wednesday in the New England Journal of Medicine.
“The data presented here include those that guided our decision to advance BNT162b2 at the 30-μg dose level to the phase 2–3, international trial to evaluate its safety and efficacy in participants 18 to 85 years of age," study authors wrote.
CLICK HERE FOR FULL CORONAVIRUS COVERAGE
The jabs produced similar antibody responses, but BNT162b2 showed to have a "milder systemic reactogenicity profile...particularly in older adults," authors wrote.
Meanwhile, Pfizer kicked off its coronavirus vaccination trials in teens aged 16-17 earlier this week. Dr. Robert Frenck is leading the trials for kids and adults at Cincinnati Children’s Hospital, where he is the director of the vaccine research center and medical director of the division of infectious diseases.
Frenck told Fox News the overall trial involves 44,000 participants of all ages (12 to 85), with a goal to enroll 600 teens aged 16 to 17.
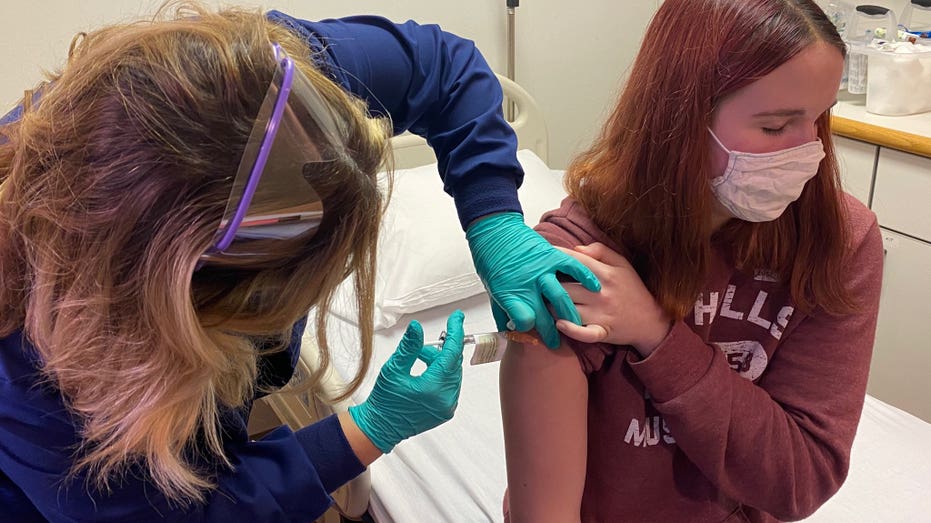
A 16-year-old patient receives a dose of Pfizer's coronavirus vaccine candidate at the Cincinnati Children's Hospital. (Photo courtesy of Cincinnati Children’s)
He also noted recent approval from the Food and Drug Administration to drop the age of allowed participants to begin testing in kids as young as 12, with a goal to enroll about 2,000 kids aged 12 to 15.
Clinical trials with a greater than minimal risk have to prove direct benefit to the child, Frenck explained, which required an accumulation of data on the vaccine’s safety profile first.
“We now have the necessary information about the safety and immune response in adults so we think it is the appropriate time to include adolescents,” he wrote in a statement to Fox News.
CLICK HERE TO GET FOX BUSINESS ON THE GO




















