CDC approves updated COVID-19 vaccines targeting omicron variant
The omicron variant is now responsible for almost all new coronavirus cases
Coronavirus booster shots may have to be taken annually: Dr. Marc Siegel
Fox News medical contributor Dr. Marc Siegel provides insight into coronavirus booster shots.
Centers for Disease Control and Prevention Director Rochelle Walensky gave the final sign off for updated COVID-19 vaccines targeting the omicron strain on Thursday.
BA.5, a subvariant of omicron, is responsible for about 89% of new coronavirus infections, according to CDC data.
The new shots, made by Pfizer and Moderna, are tweaked versions that offer half the original concoction and half to target BA.5 and BA.4.
"They can help restore protection that has waned since previous vaccination and were designed to provide broader protection against newer variants," Walensky said in a statement Thursday.

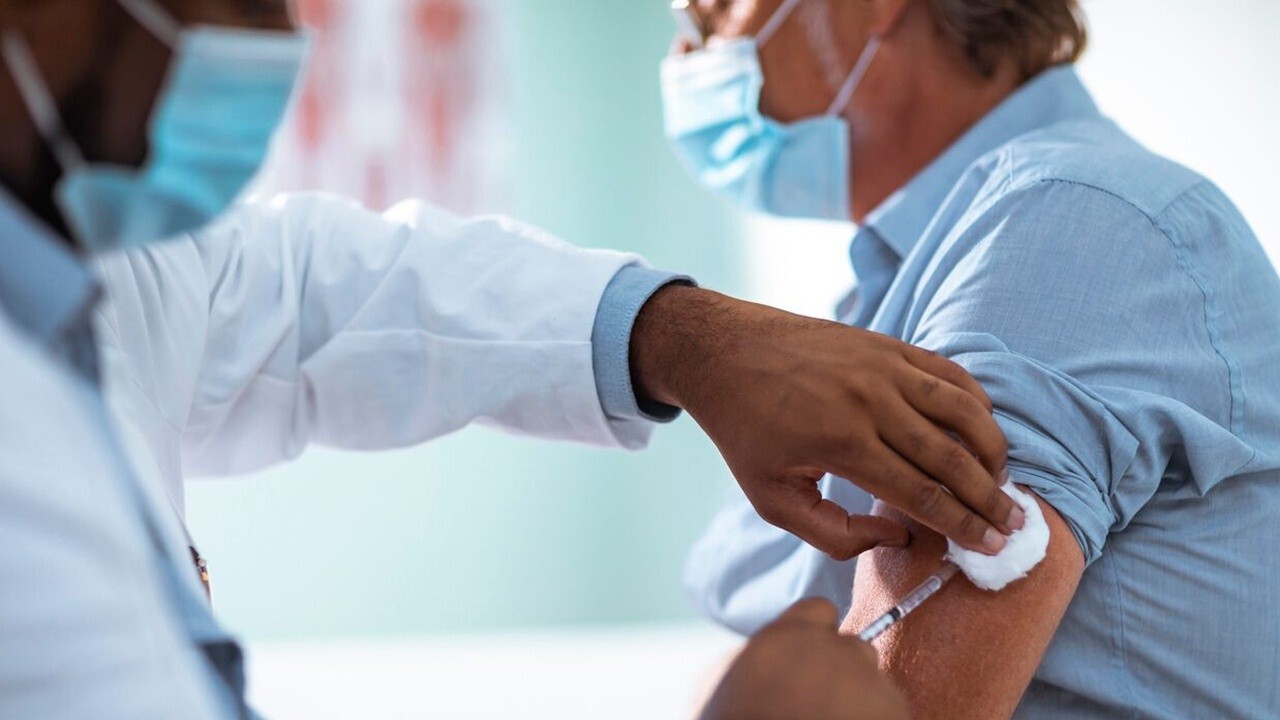
A Jackson, Miss., resident receives a Pfizer booster shot from Linda Glenn, Jackson-Hinds Comprehensive Health Center nurse, at a vaccination station next to Jackson State University in Jackson, Miss. (AP Photo/Rogelio V. Solis / AP Newsroom)
COVID-19 cases have steadily declined since mid-July, with the 7-day moving average falling from 130,387 new cases on July 16 to 85,761 on Aug. 31.
NOVAVAX TUMBLES 31% AS WANING COVID-19 VACCINE DEMAND HITS REVENUE FORECAST
Still, many Americans have resisted booster shots since they were first approved late last year. Just 51.5% of adults who received the two primary doses have received their first booster shot, according to the CDC. Only about one-third of people ages 50 and older who are eligible for a second booster shot have received that dose.
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| PFE | PFIZER INC. | 25.68 | -0.37 | -1.42% |
| MRNA | MODERNA INC. | 54.35 | +5.81 | +11.97% |
Walensky's final approval on Thursday came after the FDA and an advisory panel to the CDC recommended the new booster shots this week.
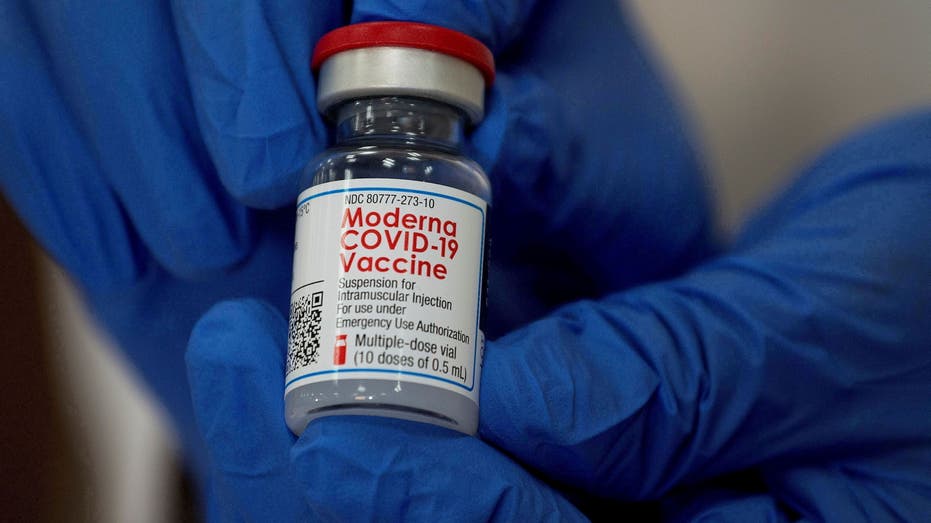
An employee shows the Moderna COVID-19 vaccine at Northwell Health's Long Island Jewish Valley Stream hospital in New York. (REUTERS/Eduardo Munoz/File Photo / Reuters Photos)
GET FOX BUSINESS ON THE GO BY CLICKING HERE
Moderna's updated vaccine was approved for people ages 18 and older, while Pfizer's was approved for people ages 12 and older.
The Department of Health and Human Services has already purchased 171 million doses of the omicron-specific vaccine.