This Marijuana Company's in Hot Water With the DOJ
Ongoing investigations into the marketing of its once high-flying fentanyl spray, Subsys, have taken a lot of the luster off Insys Therapeutics' (NASDAQ: INSY) attempt to reshape marijuana's use as medicine. Today, investigations by the Justice Department led to the arrest of former Insys Therapeutics employees, including former CEO Michael Babich, casting more uncertainty on this company's future.
Image source: Getty Images.
Troubling times
Subsys is the market-leading immediate-release transmucosal fentanyl sold in the U.S., but sales of the spray are tumbling amid revelations of off-label marketing, and growing concerns over opioid abuse.
It wasn't all that long ago that CEO Michael Babich was touting Subsys' incredible sales growth and the company's burgeoning opportunity in medical marijuana via Syndros, a reformulation of the decades-old THC drug Marinol.
Nowadays, the news surrounding Insys Therapeutics is far less optimistic.
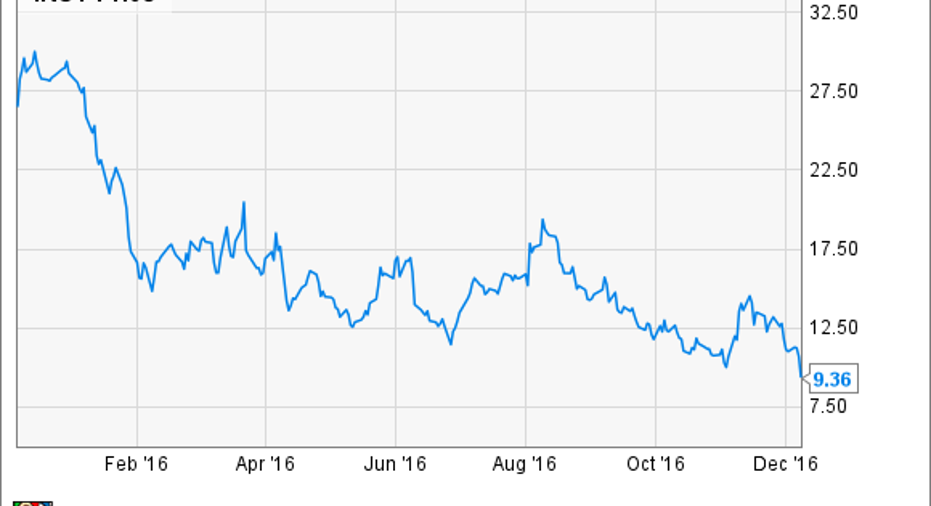
Despite winning FDA approval of Syndros earlier this year for use in chemotherapy-induced nausea and vomiting, and for anorexia in AIDS patients, Subsys' woes have made this company one of the worst-performing marijuana-related stocks:
Worry that investigations into Subsys would take an additional toll on investors was confirmed today when when the Department of Justice arrested several pharmaceutical executives and managers formerly employed by Insys Therapeutics, "on charges that they led a nationwide conspiracy to bribe medical practitioners to unnecessarily prescribe a fentanyl-based pain medication and defraud healthcare insurers."
In addition to arresting Babich, who left Insys Therapeutics as CEO and president late last year, the Justice Department arrested Insys Therapeutics' former VP of sales, national director of sales, and two regional sales directors. Those who were arrested are facing charges of bribery and illegal kickbacks to pain doctors in various states.
According to the indictment, bribes and kickbacks to prescribers were most often disguised as fees paid to the practitioners for marketing events.
The Justice Department also alleges that the defendants conspired to mislead and defraud health insurance companies. Today's indictment alleges that the defendants directed Insys Therapeutics employees to defraud insurers by "disguising the identity and location of their employer, and by lying about patient diagnosis, the type of pain being treated, and the patient's course of treatment with other medication."
In Babich's case, he's specifically being "charged with conspiracy to commit racketeering, conspiracy to commit wire and mail fraud and conspiracy to violate the Anti-Kickback Law."
Ongoing uncertainty
Insys Therapeutics had high hopes for Syndros. The company has forecast that Syndros' improved dosing flexibility and bioavailability could allow it to capture a big share of the existing $200 million Marinol market. However, the company is still awaiting Drug Enforcement Administration scheduling of Syndros, so it's not clear how much, if any, of this market Syndros will ultimately win.
In addition to predicting Syndros' success, the company has also outlined plans to develop epilepsy medicine consisting of purified cannabidiol (CBD), a non-psychoactive cannabinoid. Unfortunately, the distraction caused by the Subsys debacle makes it unclear when results from its CBD studies might be available for industry-watchers to digest.
Insys Therapeutics' shares have also been weighed down by uncertainty regarding Subsys' future sales pace, and the pending departure of founder John Kapoor, who stepped in when Babich left, but has recently said he too will be leaving.
Overall, Insys Therapeutics' troubles are a win for competitors that are developing marijuana medicine, notably GW Pharmaceuticals (NASDAQ: GWPH). GW Pharmaceuticals has reported success for its own CBD drug, Epidiolex, in three separate epilepsy trials this year, and hopes to file for FDA approval of Epidiolex next year.
As for Insys Therapeutics, it's anyone's guess whether more arrests are coming, or if the company will face fines. And for those reasons, this is one of the riskiest marijuana stocks out there.
10 stocks we like better than Insys Therapeutics When investing geniuses David and Tom Gardner have a stock tip, it can pay to listen. After all, the newsletter they have run for over a decade, Motley Fool Stock Advisor, has tripled the market.*
David and Tom just revealed what they believe are the 10 best stocks for investors to buy right now... and Insys Therapeutics wasn't one of them! That's right -- they think these 10 stocks are even better buys.
Click here to learn about these picks!
*Stock Advisor returns as of Nov. 7, 2016
Todd Campbellowns shares of Insys Therapeutics. Todd owns E.B. Capital Markets, LLC. E.B. Capital's clients may have positions in the companies mentioned. Like this article? Follow him on Twitter where he goes by the handle @ebcapital to see more articles like this.
The Motley Fool has no position in any of the stocks mentioned. Try any of our Foolish newsletter services free for 30 days. We Fools may not all hold the same opinions, but we all believe that considering a diverse range of insights makes us better investors. The Motley Fool has a disclosure policy.